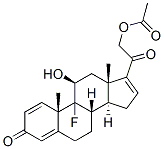
Pregna-1,4,16-triene-3,20-dione,21-(acetyloxy)-9-fluoro-11-hydroxy-, (11b)-
9-fluoro-11beta,21-dihydroxypregna-1,4,16-triene-3,20-dione 21-acetate
CAS: 1250-85-7
Molecular Formula: C23H27FO5
Pregna-1,4,16-triene-3,20-dione,21-(acetyloxy)-9-fluoro-11-hydroxy-, (11b)- - Names and Identifiers
Pregna-1,4,16-triene-3,20-dione,21-(acetyloxy)-9-fluoro-11-hydroxy-, (11b)- - Physico-chemical Properties
| Molecular Formula | C23H27FO5 |
| Molar Mass | 402.46 |
| Density | 1.28±0.1 g/cm3(Predicted) |
| Melting Point | 215-220 °C |
| Boling Point | 550.1±50.0 °C(Predicted) |
| Physical and Chemical Properties | Chemical properties crystallization. Melting point 220 ℃. |
| Use | The use of the intermediate for the determination of the acetate. |
Pregna-1,4,16-triene-3,20-dione,21-(acetyloxy)-9-fluoro-11-hydroxy-, (11b)- - Nature
| acidity coefficient (pKa) | 13.13±0.70(Predicted) |
Last Update:2024-04-10 22:29:15
Pregna-1,4,16-triene-3,20-dione,21-(acetyloxy)-9-fluoro-11-hydroxy-, (11b)- - Production method
11 α,17 α,21-trihydroxypregnoline-4-ene-3, 20-dione, that is, epihydrocortisone, can be acetylated by (21-hydroxy) and (11-hydroxy) sulfonylated and eliminated to generate 17 α,21-dihydroxypregnoline-4, 9(11) diene -3,20-diketone -21-diacetate, then it is acetylated by (17-hydroxyl group), 9[(11)-double bond] hypobromous acid addition and epoxidation, (hydrogen fluoride on the epoxy group) addition (9-fluoro, 11-position Formation of hydroxyl group) dehydrogenation (formation of 4-position double bond), elimination (16,17-position deacetate to form double bond) to obtain this product.
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: 9-fluoro-11beta,21-dihydroxypregna-1,4,16-triene-3,20-dione 21-acetate Request for quotation
CAS: 1250-85-7
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1250-85-7
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 9-fluoro-11beta,21-dihydroxypregna-1,4,16-triene-3,20-dione 21-acetate Visit Supplier Webpage Request for quotation
CAS: 1250-85-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1250-85-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 9-fluoro-11beta,21-dihydroxypregna-1,4,16-triene-3,20-dione 21-acetate Request for quotation
CAS: 1250-85-7
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1250-85-7
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: 9-fluoro-11beta,21-dihydroxypregna-1,4,16-triene-3,20-dione 21-acetate Visit Supplier Webpage Request for quotation
CAS: 1250-85-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1250-85-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

